Category: STEM (Science, Technology, Engineering and Mathematics)
ORIGINAL
Biocalcification of Sandy Gypseous Soil by Bacillus Pasteurii
Biocalcificación de suelos gipsícolas arenosos por Bacillus Pasteurii
Hadeel S. Sulaiman1 *, Muayad A. Al-Sharrad1 *, Idham A. Abed2 *
1Department of Civil Engineering, University of Anbar, Ramadi, Iraq
2College of Agriculture, University of Anbar, Ramadi, Iraq
Cite as: Sulaiman HS, Al-Sharrad MA, Abed IA. Biocalcification of Sandy Gypseous Soil by Bacillus Pasteurii. Salud, Ciencia y Tecnología - Serie de Conferencias. 2024;3: 818 https://doi.org/10.56294/sctconf2024818
Submitted: 19-01-2024 Revised: 01-04-2024 Accepted: 01-06-2024 Published: 02-06-2024
Editor: Dr.
William Castillo-González ![]()
Note: Paper presented at the 3rd Annual International Conference on Information & Sciences (AICIS’23).
ABSTRACT
Microbial-induced carbon precipitation (MICP) is one of the most recent treatment methods for soil stabilization. The present work employs this technique in improving the collapsing behavior of sandy gypseous soil with 35 % gypsum content under one-dimensional loading to 100 kPa and leaching conditions. A bacterial strain, Bacillus pasteurii was used for this purpose. A set of collapse tests was performed inside a modified oedometer on specimens, prepared with 25 % bacterial solution and 0, 0,25M, 0,5M, or 1M cementation solution molarities, cured to 7, 14, or 21 days. The results indicated that the bacterium was able to produce a considerable amount of calcium carbonate ranging from 3 % to 15 %. This carbonate was also observed by microscopic imaging of the specimens at the interparticle contacts and also on the surfaces of soil grains. Consequently, the soil gained additional bonding and the voids became smaller. Therefore, the soil became more resistant to water flow and leaching, where the treated specimen maintained nearly the same permeability with the progression of leaching, unlike the untreated specimen which showed a 7-fold increase over the same water flow condition. Over the examined load, the MICP treatment provided almost no change in the strains caused by external loading, unlike the wetting strains which exhibited a considerable reduction of 11 % to 80 %. The results of leaching strains appeared to be sensitive to the rate of flow and the cementation solution molarity.
Keywords: MICP; Sandy Gypseous Soil; Bacillus Pasteurii.
RESUMEN
La precipitación de carbono inducida por microbios (MICP) es uno de los métodos de tratamiento más recientes para la estabilización de suelos. En el presente trabajo se emplea esta técnica para mejorar el comportamiento de colapso de un suelo gipsoso arenoso con 35 % de contenido de yeso bajo carga unidimensional a 100 kPa y condiciones de lixiviación. Para ello se utilizó una cepa bacteriana, Bacillus pasteurii. Se realizó un conjunto de ensayos de colapso dentro de un edómetro modificado sobre probetas, preparadas con un 25 % de solución bacteriana y molaridades 0, 0,25M, 0,5M, o 1M de solución de cementación, curadas a 7, 14, o 21 días. Los resultados indicaron que la bacteria era capaz de producir una cantidad considerable de carbonato cálcico que oscilaba entre el 3 % y el 15 %. Este carbonato también se observó mediante imágenes microscópicas de las muestras en los contactos interpartículas y también en las superficies de los granos de suelo. En consecuencia, el suelo ganó adherencia adicional y los huecos se hicieron más pequeños. Por lo tanto, el suelo se hizo más resistente al flujo de agua y a la lixiviación, donde el espécimen tratado mantuvo casi la misma permeabilidad con la progresión de la lixiviación, a diferencia del espécimen no tratado que mostró un aumento de 7 veces sobre la misma condición de flujo de agua. Sobre la carga examinada, el tratamiento MICP no proporcionó casi ningún cambio en las deformaciones causadas por la carga externa, a diferencia de las de humectación que mostraron una reducción considerable del 11 % al 80 %. Los resultados de las deformaciones por lixiviación parecieron ser sensibles a la velocidad de flujo y a la molaridad de la solución de cementación.
Palabras clave: MICP; Suelo Gipsoso Arenoso; Bacillus Pasteurii.
INTRODUCTION
Gypseous soils pose a significant challenge for geotechnical engineers due to their complex nature. Structures or dams supported by gypseous soil are prone to unpredictable deformations that could ultimately lead to catastrophic failure. In Iraq, several structures have experienced various types of cracks and uneven deformations caused primarily by exposure to water. Gypseous soils have high bearing capacity and low compressibility in their dry state. However, when the different salts contained in the soil dissolve, new pores are created, and the bonds between the soil particles loosen. This process creates a meta-stable structure that makes it easier for the particles to slide into a denser state. The rate of gypsum dissolution depends on environmental factors such as moisture content, groundwater table and/or surface water fluctuation, climate changes such as temperature, permeability, and state of flow conditions, as well as the type and content of gypsum.(1,2,3,4,5)
Microbial-induced carbonate precipitation (MICP) is a highly promising technology for soil stabilization. This technique involves using urease-producing bacteria, which are mixed with a cementation solution and injected or blended with the soil. Several studies have found that MICP is an effective method for soil stabilization, including research by (6,7,8,9,10,11). Induced calcium carbonate is created by the hydrolysis of urea using the enzyme urease produced by the bacterium pasteurii.(7,12,13) This process, known as microbial-induced calcium carbonate precipitation (MICP), is being explored for various applications, such as soil improvement,(4,15) concrete remediation,(16,17) heavy metal removal,(18) and resistance to wind erosion(19,20,21) and construction material development.(22)
Different strains of bacteria have proven to be effective in facilitating calcite precipitation. Bacillus megaterium and pasteurii are two of the most commonly used bacterial strains due to their unique properties, such as their ability to withstand harsh environmental conditions. For instance, Bacillus pasteurii is capable of producing spores, which enables it to survive extreme conditions like high temperatures, exposure to hydrocarbon pollutants, and even extended periods of drought.(23) Bacterial strains such as Bacillus megaterium and pasteurii have also been shown to be effective in aiding calcite precipitation. These strains have unique properties, such as their ability to withstand harsh environmental conditions. In the MICP process, urea undergoes hydrolysis by microbial urease, forming NH4+ and CO32–. The produced CO32– reacts with Ca2+ to form CaCO3, which can serve as a cementation material to bind the geomaterials together, thereby increasing the material’s strength and stiffness.(24)
A microbial grouting method is widely used in the study of MICP-reinforced sand soils to obtain high-strength construction materials.(25,26,27) When using the grouting method for reinforced sand, the bacterial and cementation solutions are injected separately into molds containing loose sand samples. However, studies have shown that the grouting rate should not be lower than the rate of calcite formation, otherwise clogging would occur and much higher grouting pressure would be anticipated to advance the grout through the decreasing pores.(24) This can cause calcium carbonate crystals to accumulate and block the grouting point, making it difficult to inject the cementation solution uniformly into the soil. This can result in a non-uniform distribution of cementation in the soil, which can impact its mechanical and hydrologic properties. The grouting pressure is another important parameter that affects the mechanical and hydrologic properties of MICP-treated soils. If the pressure is excessive, it can lead to internal soil erosion, which can compromise the original soil structure and reduce the ultimate strength and permeability of the soil.(28)
The study aims to improve gypseous soil by mixing it with bacteria suspension and soaking it in a cementation solution. This method takes advantage of the dissolution of gypsum, which becomes a source of calcium ions and is important in the formation of calcite in the MICP technology.
METHODS AND MATERIALS
Soil
In this work, disturbed soil brought from a local site within the main campus of Anbar University was used. The soil is classified (ASTM D2487-17) (29) as a non-plastic poorly graded sand with silt and high gypsum content (35 %). The soil constituted of 2,3 %, 91,7 %, and 6 % gravel, sand, and fines, respectively, as determined by dry sieving with the ASTM D422-63(2007).(30) The maximum dry unit weight and the optimum moisture content, were 15,27 kN/m3 and 9,5 %, as determined with the standard Proctor compaction (ASTM D698-12).(31)
Culture medium and bacterial suspension
For this work, Bacillus pasteurii PTCC 1645 was used as the bacterium. The strain was initially preserved in glycerol at a temperature of 4°C. To prepare agar plates, 2g yeast extract, 1g (NH4)2SO4, and 2g agar were added to 100 ml of (pH 9) Tris buffer, and the solution was autoclaved at 121°C and a pressure of 15 Ib/in2 after being cooled and poured into Petri dishes to solidify. The activated isolates were cultured by striking a fresh petri dish (figure1). Subsequently, the dishes were placed in an incubator at 30°C for 48 hours.
Figure 1. Cultured bacteria in a Petri dish
The liquid media was prepared by autoclaving 1L (pH 9,0) Tris buffer, 10g (NH4)2SO4, and 20 g yeast extract at 121°C and a pressure of Ib/in2 before use. A similar procedure can be found in (32). A single colony from the surface of the agar plate with the highest concentration was cultured in 100 ml of this solution. Afterward, the flask was shaken in a shaking incubator at 180 rpm and 30°C for 48 hours. Then, the bacterial suspension was prepared at a ratio of 1:100. The concentration of the bacterial suspension was measured with a spectrophotometer at 600 nm wavelength, OD600, and was equal to 2,550. The concentration of the bacterial suspension was calculated by using equation 1 from (33) as cited in (34).
Y=8,59×107 ×Z1,3627. (1)
where Z is the reading corresponding to 600 nm wavelength, and Y represents the concentration of cells per milliliter.
Referring to equation 1, the bacterial suspension’s concentration in this work was found to be 3,076 *108 cells/ml.
Urease activity in the bacterial isolate was tested by visual inspection of the Petri dish containing urea agar, following the work described by (35,36). A color change from yellow to pink was observed, indicating a change in pH due to the hydrolysis of urea by the enzyme urease. The amount of hydrolyzing enzyme and the accumulation of ammonia were responsible for this change.
Cementation Solution
The cementation solution was prepared by mixing calcium chloride (CaCl2) and urea (CO(NH2)2) in 0,25M, 0,5M, and 1M concentrations, as recommended by (37,38). The solution was then further supplemented with 2,12 g of sodium bicarbonate (NaHCO3), 10 g of ammonium chloride (NH4Cl), and 3 g of nutrient broth per liter of deionized water.
Collapsibility and Permeability Tests
In this study, the effect of calcium carbonate on the loading, wetting, and leaching collapse of the gypseous soil was measured through a one-dimensional collapse test using a modified oedometer equipment as shown in figure 2. The oedometer cell was modified such that water flow under a constant head can be performed. Because of the corrosive effects of the chemicals included in the bacterial suspension and the cementation solution, the original oedometer ring was substituted in this work with another one made of PVC plastic (5,32 cm in diameter and 2,04 cm in height). Plastic filter discs were also manufactured for the same reason. Distilled water was used for soaking and leaching of the specimens, which was provided from a fixed head water tank. The leachate was collected from a PTFE tube connected to the outlet of the cell. The coefficient of permeability of the material was measured frequently by performing a constant head test. The variation of the leachate properties was monitored by measuring total dissolved solids TDS, and EC with a digital water tester model AZ86031.

Figure 2. Collapsibility test equipment: a) modified oedometer cell. b) test setup
Sample Preparation
As a first step, the soil was dried at a temperature of 35 oC to preserve the natural gypsum composition of the soil. Next, the dried soil was sieved on No. 4 (4,75 mm) sieve, where only the portion passing that sieve was used for the lab work. A 15 ml of preprepared bacterial suspension was mixed with 60 grams of the soil sample, making 25 % by weight of the soil sample. This allowed the bacterial suspension to effectively cover the particles of the soil. The mixture was then molded gently inside the oedometer plastic ring. To ensure that the bacteria adhered to the soil particles, the specimen was left on the bench for an hour.
Meanwhile, a container with 3 liters cementation solution was prepared for each 6 specimens. Each container was equipped with an air pump to allow for the circulation of dissolved air and provide an environment favorable for calcite production and precipitation. To prevent fine soil particles from smearing, two layers of fine filter were placed on the top and bottom faces of the specimens. These specimens were finally soaked in the cementation solution for 7, 14, or 21 days. Two samples were prepared for each treatment period, one was for the collapse test and the other was for the calcite test.
Loading, Wetting, and Leaching Process
Following the treatment stage, the oedometer ring containing the specimen was placed in the cell which was subsequently mounted in its place, as shown in figure 2. The vertical pressure was applied on the specimen by deadweights at a load incremental ratio of 1 up to a soaking pressure of 100 kPa and the change of the specimen’s height was recorded correspondingly. From an engineering perspective, the soaking pressure of 100 kPa is considered moderate given the soil’s collapsing nature, otherwise, this soil would bear a few hundred kilopascals under its dry state. After the completion of the last loading increment, the soaking process was initiated and kept on for 1 day. By the end of the soaking stage, leaching was achieved through a downward flow by opening a valve installed on the line connecting the cell to the water tank. In several tests, the leaching process was terminated after collecting 2-3 liters, however, in another number of tests, it was only possible to collect 0,2 to 0,3 liter, due to extremely lower permeability, as will be shown later in this article.
The results of the collapsibility tests were interpreted in terms of effective normal stress and axial strain. The axial strain was calculated in this study as:
ɛ=(h0-h)/h0*100. (2)
where h0 is the initial height of the specimen.
The wetting strain was calculated as:
ɛc=(h1-h2)/h0*100. (3)
where h1 and h2 are the specimen’s heights before and after wetting, respectively, at a given stress level.
The leaching strain was calculated as:
ɛl=(h2-h3)/h0*100. (4)
where h3 are specimen’s height at a given level of leaching.
The coefficient of permeability, k, was obtained under constant head condition by using Darcy’s law as:
k=v/i. (5)
where v is the discharge velocity, and i is the hydraulic gradient.
Calcium Carbonate Test by Calcimeter
The calcimeter method of estimating calcium carbonate content was used in this work (ASTM D 4373 – 02).(39) This method is based on measuring the emitted carbon dioxide gas, produced from the reaction of the calcium carbonate with the hydrochloric acid, as given below.
CaCO3+2HCL →CaCL2+H2O+CO2. (6)
The process involved mixing 0,2, 0,4, 0,6, 0,8, and 1g of calcium carbonate (99 % purity) with 50 ml of 5 % HCL acid and measuring the corresponding pressures generated from the emitted carbon dioxide gas. The results statistically analyzed and a calibration relationship was developed between the measured pressure values and the corresponding masses of the calcium carbonate. Untreated and treated soil samples, 2g each, obtained from the middle and the edge of each specimen were mixed with 50 ml of 5 % HCL acid and the corresponding pressure values were measured. The calibration relationship was then utilized to obtain the mass of the calcium carbonate of these samples. The calcium carbonate content of the soil was obtained as the mass percentage of the calculated calcium carbonate to the mass of the soil (i.e., 2 g).
RESULTS AND DISCUSSION
Calcium Carbonate Test
Figure 3 shows the results of the calcium carbonate content test on soil specimens treated with 0,25M, 0,5M, or 1,0M cementation molarities and cured for 7, 14, or 21 days. The untreated specimen yielded about 1,1 % calcium carbonate content, which is expected for many gypseous soils. In general, the calcium carbonate content increased with increasing curing time. After 7 days of curing, the amount of calcite produced by the bacterium was between 3 % (Mix 1M) and 7 % (Mix 0,5M). It appeared that the production of calcium carbonate became less apparent after 14 days of curing. After 21 days of curing, the results of the specimens with 0,5M demonstrated the highest calcium carbonate content of 15,4 %. This amount is clearly higher than that of the specimens with 0,25M and 1M cementation solution. This suggests that the cementation solution molarity of 0,5M provides the optimal medium for calcium carbonate precipitation for the gypseous soil under investigation.
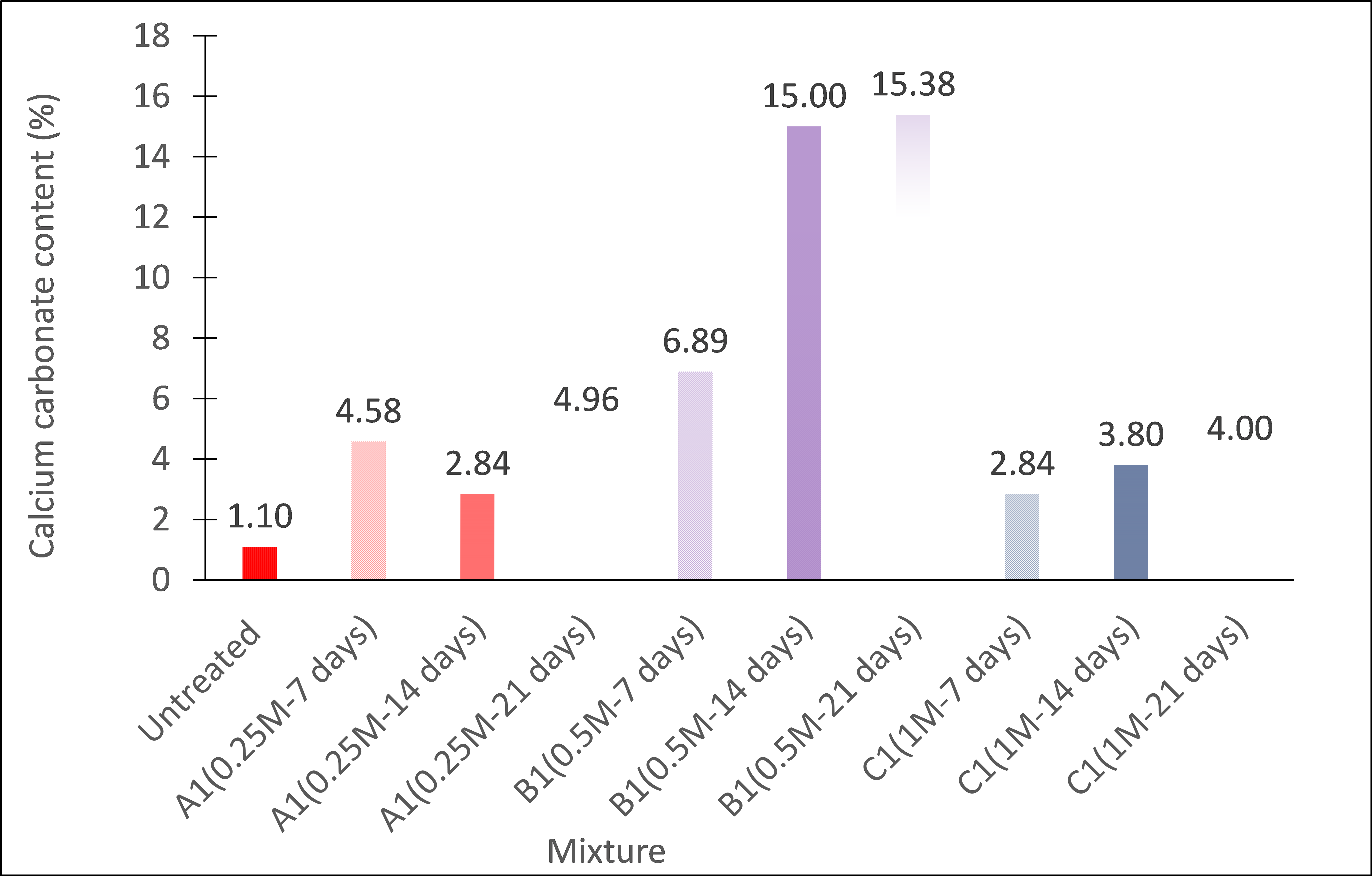
Figure 3. Calcium carbonate content of the specimens
Collapsibility Tests
Effect of Treatment on Loading and Wetting Strains
Figure 4 presents the results of the loading and wetting (soaking) stages of collapsibility tests performed on specimens treated with bacterial and cementation solutions and cured for 7, 14, or 21 days, together with the results of an untreated specimen. With the exception of the 1M specimen, the treated specimens tended to show a stiffer response (i.e., lesser strain at a given stress level) during loading, compared to the untreated specimen. The buildup of strains in these specimens was shown to be more gradual due to the progressive breakage of cementation bonding among sand particles. At the target vertical pressure, 100 kPa, most of the specimens exhibited roughly 2 % vertical strain, as shown in figure 4a.
Inspection of the results of wetting strain in figures 4, 5b, and 5c indicate that a moderate to a substantial reduction in wetting strain arose from the application of MICP treatment of the specimens with 0,5M and 1M cementation solution. For instance, the wetting strain decreased to 3,9 % and 1,7 % after 7 days of curing, and to 1,1 % and 1,4 % after 21 days, respectively. The results of the specimens with 0,25M cementation solution suggest that a minor improvement was achieved with respect to wetting strain when the specimens were cured for 7 and 21 days. Chemical inspection of the test specimens with 0,25M, indicated that more soluble chloride compounds were available in these specimens, compared to other specimens. The wetting strain for the corresponding specimen which was cured for 14 days was unexpectedly higher than that of the untreated specimen. Further inspection of this test is however anticipated.
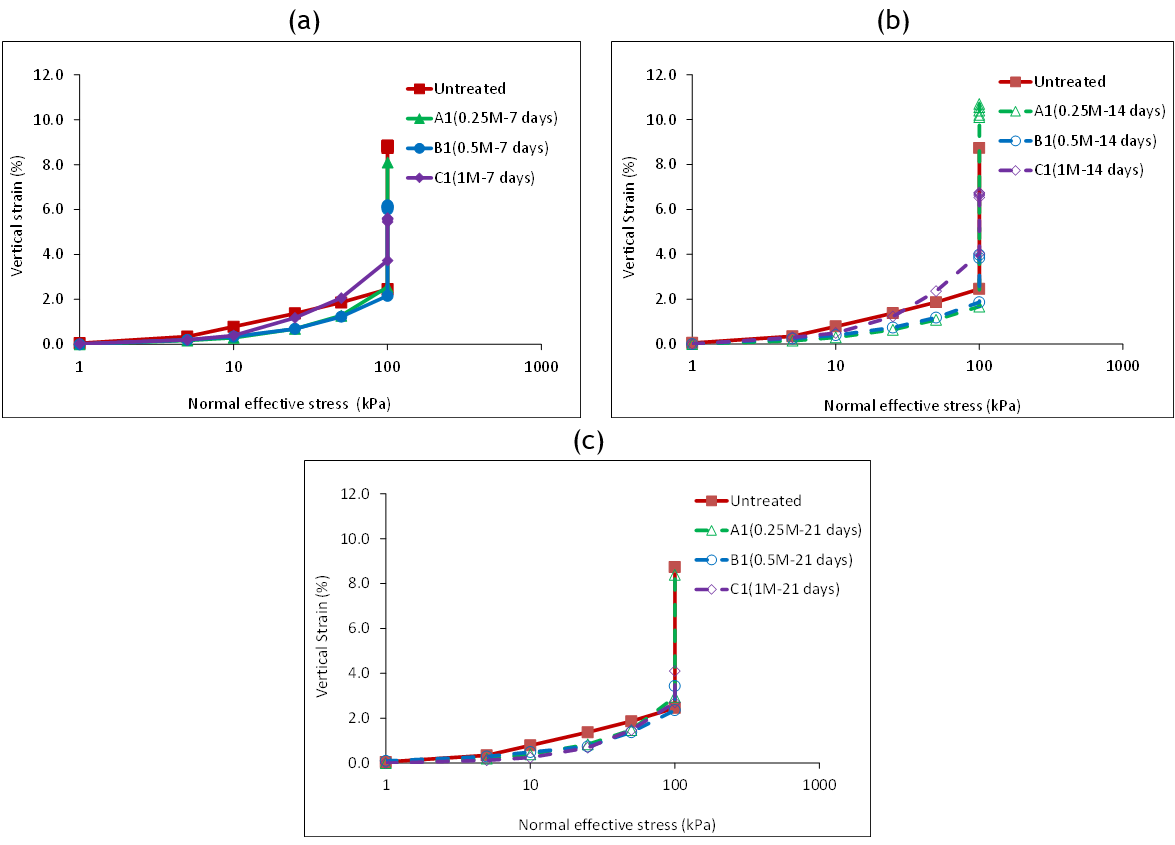
Figure 4. Effect of loading and wetting on strain: a) 7-days curing, b) 14 days curing, c)21 days curing
Effect of Treatment on Permeability
Figure 6 shows a typical variation of the coefficient of permeability with leachate volume time of leaching. With the progress of leaching, the coefficient of permeability of the untreated specimen increased by almost 7 folds. This can be attributed to gypsum leaching which appeared to cause an enlargement in the voids and water passages through the specimen. The MICP-treated specimens showed slightly lower k values at the beginning, then marginally increased with the progress of leaching. This performance is associated with the fact that the produced calcium carbonate provides cementation and coating to soil grains and gypsum in such a way that water flow has little effect on salt leaching. Similar trends can be found in.(40)

Figure 5. Effect of curing age on: a) loading strain, b) wetting strain, c) loading and wetting starin
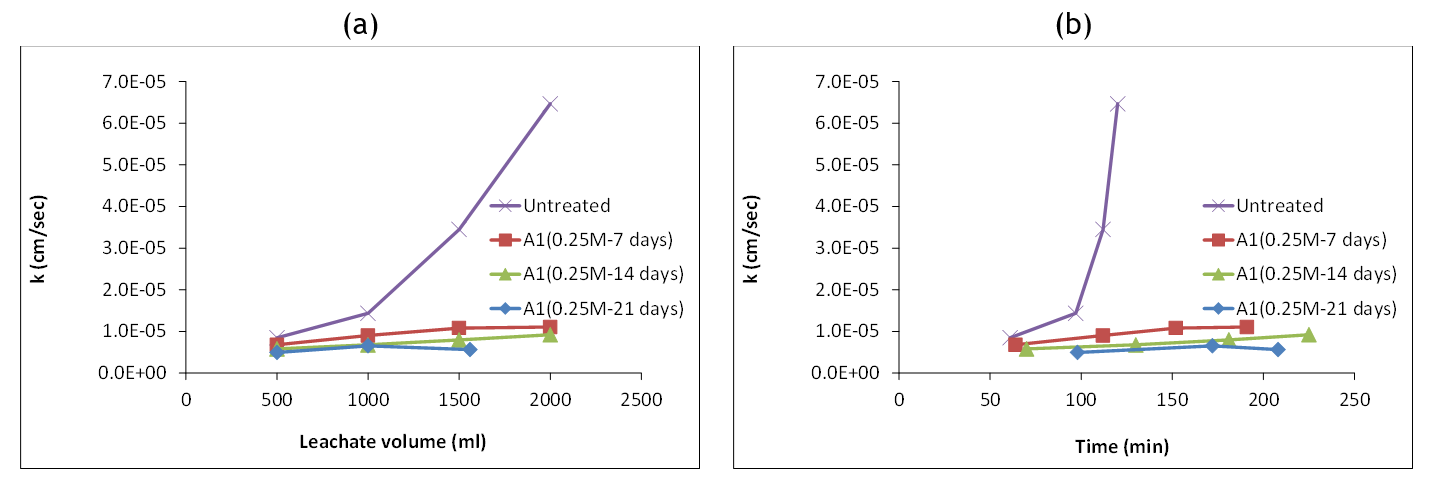
Figure 6. Typical variation of the coefficient of permeability with: a) leachate volume, b) time of leaching
Effect of Treatment on Leaching Strain
An example of the variation of leaching strain with leachate volume under a 100 kPa vertical pressure is shown in figure 7 for a specimen with 1M cementation solution subjected to 21 days of treatment. Inspection of figure 7a indicates that the leachate was discharged at the same rate over the first 100 minutes. This provides a basis for comparing the leaching strains of the two specimens over that time. Clearly, the treated specimen exhibited a larger leaching strain than the untreated one, as shown in figure 7b. The corresponding variation of the TDS is shown in figure 7c. The TDS values from the treated specimen were always higher than that from the untreated specimen. This is expected as the treated specimen has an abundance of chemical substances other than gypsum emerging from the treatment stage, which has negatively impacted the magnitude of the leaching strain. After 100 minutes, the untreated specimen experienced a significant increase in permeability. As permeability increases, water seeps more rapidly, therefore causing less gypsum dissolution and leaching. Accordingly, lesser leaching strains were recorded for that specimen compared to the treated one.
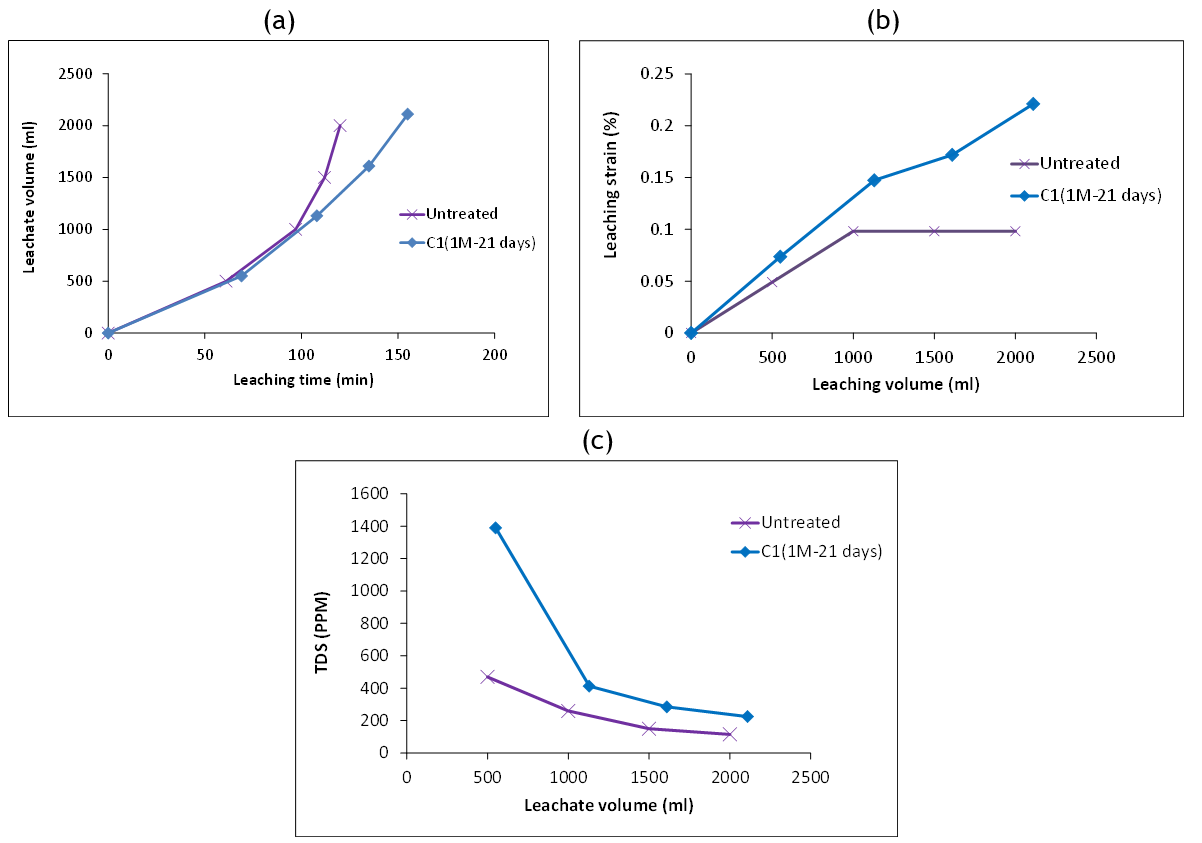
Figure 7. Variation of leachate volume with: a) leaching time, b) leaching strain, c) TDS
Microstructural and Mineralogical Analysis
Figure 8 displays two SEM images taken on an untreated specimen at magnifications of 160x and 1300x. Inspection of figure 8a demonstrates that soil structure is mainly comprised of sand particles contaminated and bonded by gypsum which appears as individual or lumped crystals. This is rather obvious in figure 8b. The material is also characterized by an interconnected network of pores as large as 710 μm.
The inclusion of calcium carbonate resulted in a number of interesting changes in the soil microstructure (figure 9). The voids of the soil were largely occupied by calcium carbonate (figure 9a), resulting in smaller voids of 150- 400 μm. The effect of decreasing void size was profound on the values of the strains caused by loading and soaking as well as on the permeability, as demonstrated earlier in figures 4 to 6. Close inspection of figure 9b indicates that the three polymorphs of calcium carbonate (i.e., calcite, aragonite, and vaterite) are coexisted within the soil, but with different abundances. Calcite, which appears to be the most abundant mineral, is characterized by trigonal crystal system, whereas aragonite and vaterite are less abundant minerals, and are characterized with orthorhombic and hexagonal crystal systems, respectively.(41)

Figure 8. SEM images of untreated specimen magnified to: a) 160x, b) 1300x

Figure 9. SEM images of MICP treated specimen (0,5M cementation solution -21 days) magnified to: a) 150x, b) 1300x
CONCLUSIONS
The current study explored the effect of MICP treatment, with Bacillus pasteurii, on the collapsing behavior of a sandy soil containing about 35 % gypsum content. Untreated and treated specimens were prepared to investigate one-dimensional collapsibility, leachate, and flow characteristics. The results demonstrated that a moderate to high calcium carbonate content of 3 % and 15 % was produced by various mixtures, with a clear correlation between calcium carbonate content and cementation solution molarity, where the calcium carbonate content peaked at 0,5M and increased with increasing curing age.
Analysis of the collapsibility and leaching tests indicated that, at the beginning of leaching, treated specimens yielded slightly lower values of the coefficient of permeability compared to the untreated specimen. The treated specimens tended to maintain the same permeability with leaching, compared to the untreated one which exhibited many times increase in permeability as a result of gypsum dissolution and leaching. The strain due to loading does not appear to be affected by treatment, despite the microstructural observation that, voids became smaller after treatment. In most of the tests, the wetting strain was effectively reduced by treatment by 11 % to 80 % with the suggestion that the higher the cementation solution molarity and the longer curing is the lower wetting strain. The leaching strain obtained in this work was shown to be affected by soil permeability where over a given leachate volume, the samples with higher permeability tend to show lesser leaching strain. This was attributed to the fact that higher discharge velocities do not allow for much gypsum dissolution.
REFERENCES
1. S. Casby-Horton, J. Herrero, and N. A. Rolong, “Gypsum soils—Their morphology, classification, function, and landscapes,” Adv. Agron., vol. 130, pp. 231–290, 2015.
2. A. Jotisankasa, “Collapse behaviour of a compacted silty clay.” University of London London, UK, 2005.
3. G. Bolzon, “Collapse mechanisms at the foundation interface of geometrically similar concrete gravity dams,” Eng. Struct., vol. 32, no. 5, pp. 1304–1311, 2010.
4. M. Y. Fattah, I. H. Obead, and H. A. Omran, “A study on leaching of collapsible gypseous soils,” Int. J. Geotech. Eng., vol. 16, no. 1, pp. 44–54, 2022.
5. S. S. Razouki and O. A. El-Janabi, “Decrease in the CBR of a gypsiferous soil due to long-term soaking,” Q. J. Eng. Geol. Hydrogeol., vol. 32, no. 1, pp. 87–89, 1999.
6. B. C. Martinez, T. H. Barkouki, J. D. DeJong, and T. R. Ginn, “Upscaling Microbial Induced Calcite Precipitation in 0.5m Columns: Experimental and Modeling Results,” vol. 41165, no. March, pp. 4049–4059, 2011, doi: 10.1061/41165(397)414.
7. A. Al Qabany, K. Soga, and C. Santamarina, “Factors affecting efficiency of microbially induced calcite precipitation,” J. Geotech. Geoenvironmental Eng., vol. 138, no. 8, pp. 992–1001, 2012.
8. A. Al Qabany and K. Soga, “Effect of chemical treatment used in MICP on engineering properties of cemented soils,” in Bio-and Chemo-Mechanical Processes in Geotechnical Engineering: Géotechnique Symposium in Print 2013, ICE Publishing, 2014, pp. 107–115.
9. L. Cheng, R. Cord-Ruwisch, and M. A. Shahin, “Cementation of sand soil by microbially induced calcite precipitation at various degrees of saturation,” Can. Geotech. J., vol. 50, no. 1, pp. 81–90, 2013.
10. Z. S. Hadi and K. A. Saeed, “Effect of microbial-induced calcite precipitation (MICP) on the strength of soil contaminated with lead nitrate,” J. Mech. Behav. Mater., vol. 31, no. 1, pp. 143–149, 2022, doi: 10.1515/jmbm-2022-0016.
11. A. D. Almurshedi and M. Karkush, “Experimental and numerical modeling of load settlement behavior of gypseous soils improved by MICP,” in Smart Geotechnics for Smart Societies, CRC Press, 2023, pp. 583–589.
12. N. K. Dhami, M. S. Reddy, and A. Mukherjee, “Biomineralization of calcium carbonate polymorphs by the bacterial strains isolated from calcareous sites,” J. Microbiol. Biotechnol., vol. 23, no. 5, pp. 707–714, 2013.
13. A. D. Salman, M. O. Karkush, and H. H. Karim, “Effect of microbial induced calcite precipitation on shear strength of gypseous soil in dry and soaking conditions,” in Geotechnical Engineering and Sustainable Construction: Sustainable Geotechnical Engineering, Springer, 2022, pp. 103–114.
14. B. C. Martinez et al., “Experimental optimization of microbial-induced carbonate precipitation for soil improvement,” J. Geotech. Geoenvironmental Eng., vol. 139, no. 4, pp. 587–598, 2013.
15. S. Liu, K. Du, K. Wen, W. Huang, F. Amini, and L. Li, “Sandy soil improvement through microbially induced calcite precipitation (MICP) by immersion,” J. Vis. Exp., vol. 2019, no. 151, 2019, doi: 10.3791/60059.
16. X. Sun, L. Miao, L. Wu, and H. Wang, “Theoretical quantification for cracks repair based on microbially induced carbonate precipitation (MICP) method,” Cem. Concr. Compos., vol. 118, p. 103950, 2021.
17. Z. S. Hadi and K. A. Saeed, “Effect of microbial-induced calcite precipitation (MICP) on the strength of soil contaminated with lead nitrate,” J. Mech. Behav. Mater., vol. 31, no. 1, pp. 143–149, 2022.
18. H. Bai et al., “Microbially-induced calcium carbonate precipitation by a halophilic ureolytic bacterium and its potential for remediation of heavy metal-contaminated saline environments,” Int. Biodeterior. Biodegradation, vol. 165, p. 105311, 2021.
19. Z. Wang, N. Zhang, J. Ding, C. Lu, and Y. Jin, “Experimental study on wind erosion resistance and strength of sands treated with microbial-induced calcium carbonate precipitation,” Adv. Mater. Sci. Eng., vol. 2018, 2018.
20. F. Nikseresht, A. Landi, G. Sayyad, G. R. Ghezelbash, and R. Schulin, “Sugarecane molasse and vinasse added as microbial growth substrates increase calcium carbonate content, surface stability and resistance against wind erosion of desert soils,” J. Environ. Manage., vol. 268, p. 110639, 2020.
21. H. Meng, S. Shu, Y. Gao, J. He, and Y. Wan, “Kitchen waste for Sporosarcina pasteurii cultivation and its application in wind erosion control of desert soil via microbially induced carbonate precipitation,” Acta Geotech., vol. 16, no. 12, pp. 4045–4059, 2021.
22. S. E. Lambert and D. G. Randall, “Manufacturing bio-bricks using microbial induced calcium carbonate precipitation and human urine,” Water Res., vol. 160, pp. 158–166, 2019.
23. L. Wang, T. L. K. Yeung, A. Y. T. Lau, D. C. W. Tsang, and C.-S. Poon, “Recycling contaminated sediment into eco-friendly paving blocks by a combination of binary cement and carbon dioxide curing,” J. Clean. Prod., vol. 164, pp. 1279–1288, 2017.
24. Y. Wang, G. Wang, Y. Wan, X. Yu, J. Zhao, and J. Shao, “Recycling of dredged river silt reinforced by an eco-friendly technology as microbial induced calcium carbonate precipitation (MICP),” Soils Found., vol. 62, no. 6, p. 101216, 2022.
25. D. J. Tobler, E. Maclachlan, and V. R. Phoenix, “Microbially mediated plugging of porous media and the impact of differing injection strategies,” Ecol. Eng., vol. 42, pp. 270–278, 2012.
26. A. Almajed, H. K. Tirkolaei, E. Kavazanjian, and N. Hamdan, “Enzyme induced biocementated sand with high strength at low carbonate content. Sci Rep 9: 1135.” 2019.
27. B. Liu et al., “Potential drought mitigation through microbial induced calcite precipitation‐MICP,” Water Resour. Res., vol. 57, no. 9, p. e2020WR029434, 2021.
28. B. Liu et al., “Bio-remediation of desiccation cracking in clayey soils through microbially induced calcite precipitation (MICP),” Eng. Geol., vol. 264, p. 105389, 2020.
29. “ASTM D2487-17 Standard Practice for Classification of Soils for Engineering Purposes (Unified Soil Classification System) ASTM International, West Conshohocken, PA (2017), 10.1520/D2487-17”.
30. ASTM D422. (2007). Standard test method for particle-size analysis of soils (ASTM International). www.astm.orgNo Title.
31. ASTM D698. (2012). Standard test method for laboratory compaction characteristics of soil using standard effort (12 400 ft-lbf/ft3 (600 kN-m/m3)). (ASTM International). www.astm.orgNo Title.
32. Y. Mo, S. Yue, Q. Zhou, and X. Liu, “Improvement and soil consistency of sand–clay mixtures treated with enzymatic-induced carbonate precipitation,” Materials (Basel)., vol. 14, no. 18, 2021, doi: 10.3390/ma14185140.
33. S. K. Ramachandran, V. Ramakrishnan, and S. S. Bang, “Remediation of concrete using microorganisms,” Mater. J., vol. 98, no. 1, pp. 3–9, 2001.
34. W. Wan et al., “Isolation and characterization of phosphorus solubilizing bacteria with multiple phosphorus sources utilizing capability and their potential for lead immobilization in soil,” Front. Microbiol., vol. 11, p. 752, 2020.
35. V. Achal, A. Mukherjee, P. C. Basu, and M. S. Reddy, “Strain improvement of Sporosarcina pasteurii for enhanced urease and calcite production,” J. Ind. Microbiol. Biotechnol., vol. 36, no. 7, pp. 981–988, 2009.
36. J. G. Collee, Mackie & McCartney practical medical microbiology., 14th ed. / edited... Edinburgh ; Churchill Livingstone, 1996.
37. Wei-Soon Ng, Min-Lee Lee, and Siew-Ling Hii, “An Overview of the Factors Affecting Microbial-Induced Calcite Precipitation and its Potential Application in Soil Improvement,” Int. J. Civ. Environ. Eng., vol. 6, no. 2, pp. 188–194, 2012, Online.. Available: https://pdfs.semanticscholar.org/dc1f/0edb47ecdc6b4a1ce6b46ab6a4114ae60503.pdf
38. M. Nemati, E. A. Greene, and G. Voordouw, “Permeability profile modification using bacterially formed calcium carbonate: comparison with enzymic option,” Process Biochem., vol. 40, no. 2, pp. 925–933, 2005.
39. “ASTM D 4373 – 02_Standard Test Method for Rapid Determination of Carbonate Content of Soi”.
40. I. R. K. Phang, K. S. Wong, Y. S. Chan, and S. Y. Lau, “Effect of microbial-induced calcite precipitation towards strength and permeability of peat,” Bull. Eng. Geol. Environ., vol. 81, no. 8, 2022, doi: 10.1007/s10064-022-02790-0.
41. P. Anbu, C.-H. Kang, Y.-J. Shin, and J.-S. So, “Formations of calcium carbonate minerals by bacteria and its multiple applications,” Springerplus, vol. 5, no. 1, pp. 1–26, 2016.
FINANCINGNone. CONFLICT OF INTERESTNone. AUTHORSHIP CONTRIBUTIONConceptualization: Hadeel S. Sulaiman, Muayad A. Al-Sharrad, Idham A. Abed.
Research: Hadeel S. Sulaiman, Muayad A. Al-Sharrad, Idham A. Abed.
Writing - original draft: Hadeel S. Sulaiman, Muayad A. Al-Sharrad, Idham A. Abed.
Writing - revision and editing: Hadeel S. Sulaiman, Muayad A. Al-Sharrad, Idham A. Abed.